ELISA Kit for Interleukin 12B (IL12B) 
p40; IL12-B; CLMF2; NKSF2; Natural Killer Cell Stimulatory Factor 2; Cytotoxic Lymphocyte Maturation Factor 2; Interleukin 23, P40 Subunit
- UOM
- FOB US$ 441.00 US$ 630.00 US$ 2,835.00 US$ 5,355.00 US$ 44,100.00
- Quantity
Overview
Properties
- Product No.SEA058Hu
- Organism SpeciesHomo sapiens (Human) Same name, Different species.
- ApplicationsEnzyme-linked immunosorbent assay for Antigen Detection.
Research use only - DownloadInstruction Manual
- CategoryCytokineInfection immunityImmune molecule
Sign into your account
Share a new citation as an author
Upload your experimental result
Review

Contact us
Please fill in the blank.
Recovery
Matrices listed below were spiked with certain level of recombinant Interleukin 12B (IL12B) and the recovery rates were calculated by comparing the measured value to the expected amount of Interleukin 12B (IL12B) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 79-104 | 86 |
| EDTA plasma(n=5) | 93-101 | 98 |
| heparin plasma(n=5) | 89-96 | 93 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Interleukin 12B (IL12B) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Interleukin 12B (IL12B) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Interleukin 12B (IL12B) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 79-91% | 88-101% | 78-97% | 85-92% |
| EDTA plasma(n=5) | 96-103% | 89-103% | 94-102% | 86-93% |
| heparin plasma(n=5) | 92-104% | 92-101% | 97-105% | 78-101% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| Pre-coated, ready to use 96-well strip plate | 1 | Plate sealer for 96 wells | 4 |
| Standard | 2 | Standard Diluent | 1×20mL |
| Detection Reagent A | 1×120µL | Assay Diluent A | 1×12mL |
| Detection Reagent B | 1×120µL | Assay Diluent B | 1×12mL |
| TMB Substrate | 1×9mL | Stop Solution | 1×6mL |
| Wash Buffer (30 × concentrate) | 1×20mL | Instruction manual | 1 |
Assay procedure summary
1. Prepare all reagents, samples and standards;
2. Add 100µL standard or sample to each well. Incubate 1 hours at 37°C;
3. Aspirate and add 100µL prepared Detection Reagent A. Incubate 1 hour at 37°C;
4. Aspirate and wash 3 times;
5. Add 100µL prepared Detection Reagent B. Incubate 30 minutes at 37°C;
6. Aspirate and wash 5 times;
7. Add 90µL Substrate Solution. Incubate 10-20 minutes at 37°C;
8. Add 50µL Stop Solution. Read at 450nm immediately.

Test principle
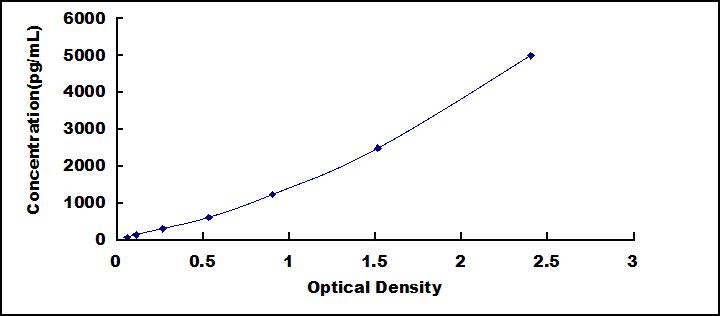
The test principle applied in this kit is Sandwich enzyme immunoassay. The microtiter plate provided in this kit has been pre-coated with an antibody specific to Interleukin 12B (IL12B). Standards or samples are then added to the appropriate microtiter plate wells with a biotin-conjugated antibody specific to Interleukin 12B (IL12B). Next, Avidin conjugated to Horseradish Peroxidase (HRP) is added to each microplate well and incubated. After TMB substrate solution is added, only those wells that contain Interleukin 12B (IL12B), biotin-conjugated antibody and enzyme-conjugated Avidin will exhibit a change in color. The enzyme-substrate reaction is terminated by the addition of sulphuric acid solution and the color change is measured spectrophotometrically at a wavelength of 450nm ± 10nm. The concentration of Interleukin 12B (IL12B) in the samples is then determined by comparing the O.D. of the samples to the standard curve.
Giveaways
Increment services
-
 Single-component Reagents of Assay Kit
Single-component Reagents of Assay Kit
-
 Lysis Buffer Specific for ELISA / CLIA
Lysis Buffer Specific for ELISA / CLIA
-
 Quality Control of Kit
Quality Control of Kit
-
 ELISA Kit Customized Service
ELISA Kit Customized Service
-
 Disease Model Customized Service
Disease Model Customized Service
-
 Serums Customized Service
Serums Customized Service
-
 TGFB1 Activation Reagent
TGFB1 Activation Reagent
-
 Real Time PCR Experimental Service
Real Time PCR Experimental Service
-
 Streptavidin
Streptavidin
-
 Fast blue Protein Stain solution
Fast blue Protein Stain solution -
 Single-component Reagents of FLIA Kit
Single-component Reagents of FLIA Kit
-
 Streptavidin-Agarose Beads
Streptavidin-Agarose Beads
Citations
- Establishment and evaluation of a bead-based luminex assay allowing simultaneous quantification of equine IL-12 and IFN-γ.Pubmed: 23564769
- Serum Proteomics Analysis in Rats of Immunosuppression Induced by Chronic Stress.pubmed:27384769
- Impact of purified oat 1-3, 1-4-β-d-glucan of different molecular weight on alleviation of inflammation parameters during gastritisS1756464616303528
- Photothermal-triggered Controlled Drug Release From Mesoporous Silica nanoparticles Based On Base-pairing Rules
- Effects of Early Intervention with Antibiotics and Maternal Fecal Microbiota on Transcriptomic Profiling Ileal Mucusa in Neonatal PigsPubmed: 31963653
- Fasciola gigantica excretory-secretory products (FgESPs) modulate the differentiation and immune functions of buffalo dendritic cells through a mechanism …Pubmed: 32680553