ELISA Kit for Visfatin (VF) 
Nampt; NAmPRTase; PBEF; PBEF1; Pre-B-Cell Colony Enhancing Factor; Nicotinamide Phosphoribosyltransferase
- UOM
- FOB US$ 466.00 US$ 665.00 US$ 2,993.00 US$ 5,653.00 US$ 46,550.00
- Quantity
Overview
Properties
- Product No.CEA638Hu
- Organism SpeciesHomo sapiens (Human) Same name, Different species.
- ApplicationsEnzyme-linked immunosorbent assay for Antigen Detection.
Research use only - DownloadInstruction Manual
- CategoryMetabolic pathwayCardiovascular biologyHepatology
Sign into your account
Share a new citation as an author
Upload your experimental result
Review

Contact us
Please fill in the blank.
Recovery
Matrices listed below were spiked with certain level of recombinant Visfatin (VF) and the recovery rates were calculated by comparing the measured value to the expected amount of Visfatin (VF) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 87-101 | 94 |
| EDTA plasma(n=5) | 94-102 | 99 |
| heparin plasma(n=5) | 82-93 | 85 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Visfatin (VF) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Visfatin (VF) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Visfatin (VF) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 91-101% | 90-101% | 86-98% | 88-104% |
| EDTA plasma(n=5) | 96-104% | 89-97% | 82-93% | 78-92% |
| heparin plasma(n=5) | 91-99% | 79-97% | 81-93% | 81-92% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| Pre-coated, ready to use 96-well strip plate | 1 | Plate sealer for 96 wells | 4 |
| Standard | 2 | Standard Diluent | 1×20mL |
| Detection Reagent A | 1×120µL | Assay Diluent A | 1×12mL |
| Detection Reagent B | 1×120µL | Assay Diluent B | 1×12mL |
| TMB Substrate | 1×9mL | Stop Solution | 1×6mL |
| Wash Buffer (30 × concentrate) | 1×20mL | Instruction manual | 1 |
Assay procedure summary
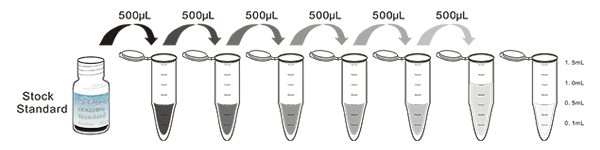
1. Prepare all reagents, samples and standards;
2. Add 50µL standard or sample to each well.
And then add 50µL prepared Detection Reagent A immediately.
Shake and mix. Incubate 1 hour at 37°C;
3. Aspirate and wash 3 times;
4. Add 100µL prepared Detection Reagent B. Incubate 30 minutes at 37°C;
5. Aspirate and wash 5 times;
6. Add 90µL Substrate Solution. Incubate 10-20 minutes at 37°C;
7. Add 50µL Stop Solution. Read at 450 nm immediately.
Test principle
This assay employs the competitive inhibition enzyme immunoassay technique. An antibody specific to visfatin has been pre-coated onto a microplate. A competitive inhibition reaction is launched between biotin labeled visfatin and unlabeled visfatin (Standards or samples) with the pre-coated antibody specific to visfatin. After incubation the unbound conjugate is washed off. Next, avidin conjugated to Horseradish Peroxidase (HRP) is added to each microplate well and incubated. The amount of bound HRP conjugate is reverse proportional to the concentration of visfatin in the sample. After addition of the substrate solution, the intensity of color developed is reverse proportional to the concentration of visfatin in the sample.
Giveaways
Increment services
Citations
- EFFECT OF 8 WEEKS ENDURANCE TRAINING ON PLASMA VISFATIN IN MIDDLE-AGED MENBrjb: source
- The relationship between visfatin and HOMA-IR in hypertensive patients, and the effect of antihypertensive drugs on visfatin and HOMA-IR in hypertensive patients with insulin resistanceScienceDirect: S0168822711002956
- Early-effect of bariatric surgery (Scopinaro method) on intestinal hormones and adipokines in insulin resistant Wistar rat.Pubmed: 24304571
- CTRP3 modulates the expression and secretion of adipokines in 3T3-L1 adipocytesPubmed:25168658
- Plasma visfatin levels and mRNA expression of visfatin in peripheral blood mononuclear cells and peripheral blood monocyte-derived macrophages from normal weight females with polycystic ovary syndromePubmed:Pmc3991522
- Visfatin derived from ascites promotes ovarian cancer cell migration through Rho/ROCK signaling-mediated actin polymerization.Pubmed:25055182
- Visfatin −948G/T and resistin −420C/Gpolymorphisms in Egyptian type 2 diabetic patients with and without cardiovascular diseasesNrcresearchpress:Source
- Antidiabetic Effect of Galantamine: Novel Effect for a Known Centrally Acting DrugPubMed: 26262991
- Changes in concentration of visfatin during four weeks of inpatient treatment of alcohol dependent malesScience: Article
- Variation in blood levels of hormones in obese patients following weight reduction induced by endoscopic and surgical bariatric therapiesPubMed: 26539806
- Genetic Variations in Visfatin and Resistin Genes in Egyptian Type 2 Diabetic Patients With and Without ComplicationsPubMed: 25120107
- Favorable outcomes of hydroxychloroquine in insulin resistance may be accomplished by adjustment of the endothelial dysfunction as well as the skewed balance of adipokinesscience:S0065128116301234
- Changes in Serum Nampt Levels and Its Significances in Diabetic Nephropathy Patients-The Potential Role of Nampt in T2DM with Diabetic Nephropathy.pubmed:28699485
- Post-chemotherapy serum anti-müllerian hormone level predicts ovarian function recoveryPubmed:30012648
- Effect of maternal hypothyroidism during pregnancy on insulin resistance, lipid accumulation and mitochondrial dysfunction in skeletal muscle of fetal ratsPubmed:29784871
- Hyperinsulinemic-euglycemic clamp over the first year of use of depot-medroxyprogesterone acetate as a contraceptivePubmed:29665358
- Analysis of synovial fluid visfatin level in temporomandibular joint disordersPubmed:29385907
- Resistin and visfatin concentrations are related to central obesity and inflammation in Brazilian children10.1186:s41110-018-0060-7
- Transcriptional Regulation of NAMPT Gene by Glycogen Synthase Kinase 3β in Goat AdipocytesPubmed: 30615481
- The Effect of 8 Weeks Aquatic Exercise Program on Plasma Visfatin Level and Some Blood Factors in Obese Men
- Adipokines as Biomarkers in Health and Disease
- Plasma concentration and expression of adipokines in epicardial and subcutaneous adipose tissue are associated with impaired left ventricular filling patternPubmed: 31533725
- Relationshıp of spontaneous subarachnoid haemorrhage and cerebral aneurysm to serum Visfatin and Nesfatin-1 levelsPubmed: 32311618
- Effects of 10-Week Concurrent Training on Insulin Resistance and the Serum Levels of Vaspin and Visfatin in Overweight Females
- Different Ratios of Corn and Coconut Oil Blends in High©\Fat Diets Influence Fat Deposition without Altering Metabolic Biomarkers in Male Rats