CLIA Kit for Ubiquitin Carboxyl Terminal Hydrolase L1 (UCHL1) 
PARK5; PGP9.5; Uch-L1; Neuron cytoplasmic protein 9.5; Ubiquitin thioesterase L1; Ubiquitin carboxyl-terminal hydrolase isozyme L1
- UOM
- FOB US$ 638.00 US$ 912.00 US$ 4,104.00 US$ 7,752.00 US$ 63,840.00
- Quantity
Overview
Properties
- Product No.SCG945Ra
- Organism SpeciesRattus norvegicus (Rat) Same name, Different species.
- ApplicationsChemiluminescent immunoassay for Antigen Detection.
Research use only - DownloadInstruction Manual
- CategoryEnzyme & Kinase
Sign into your account
Share a new citation as an author
Upload your experimental result
Review

Contact us
Please fill in the blank.
Recovery
Matrices listed below were spiked with certain level of recombinant Ubiquitin Carboxyl Terminal Hydrolase L1 (UCHL1) and the recovery rates were calculated by comparing the measured value to the expected amount of Ubiquitin Carboxyl Terminal Hydrolase L1 (UCHL1) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 85-99 | 95 |
| EDTA plasma(n=5) | 87-96 | 92 |
| heparin plasma(n=5) | 96-105 | 101 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Ubiquitin Carboxyl Terminal Hydrolase L1 (UCHL1) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Ubiquitin Carboxyl Terminal Hydrolase L1 (UCHL1) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Ubiquitin Carboxyl Terminal Hydrolase L1 (UCHL1) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 82-101% | 78-101% | 81-104% | 92-101% |
| EDTA plasma(n=5) | 86-101% | 85-95% | 89-102% | 86-101% |
| heparin plasma(n=5) | 83-101% | 85-101% | 85-92% | 87-97% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| Pre-coated, ready to use 96-well strip plate | 1 | Plate sealer for 96 wells | 4 |
| Standard | 2 | Standard Diluent | 1×20mL |
| Detection Reagent A | 1×120µL | Assay Diluent A | 1×12mL |
| Detection Reagent B | 1×120µL | Assay Diluent B | 1×12mL |
| Substrate A | 1×10mL | Substrate B | 1×2mL |
| Wash Buffer (30 × concentrate) | 1×20mL | Instruction manual | 1 |
Assay procedure summary
1. Prepare all reagents, samples and standards;
2. Add 100µL standard or sample to each well. Incubate 1 hours at 37°C;
3. Aspirate and add 100µL prepared Detection Reagent A. Incubate 1 hour at 37°C;
4. Aspirate and wash 3 times;
5. Add 100µL prepared Detection Reagent B. Incubate 30 minutes at 37°C;
6. Aspirate and wash 5 times;
7. Add 100µL Substrate Solution. Incubate 10 minutes at 37°C;
8. Read RLU value immediately.
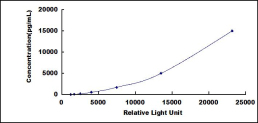
Test principle
The microplate provided in this kit has been pre-coated with an antibody specific to Ubiquitin Carboxyl Terminal Hydrolase L1 (UCHL1). Standards or samples are then added to the appropriate microplate wells with a biotin-conjugated antibody specific to Ubiquitin Carboxyl Terminal Hydrolase L1 (UCHL1). Next, Avidin conjugated to Horseradish Peroxidase (HRP) is added to each microplate well and incubated. Then the mixture of substrate A and B is added to generate glow light emission kinetics. Upon plate development, the intensity of the emitted light is proportional to the Ubiquitin Carboxyl Terminal Hydrolase L1 (UCHL1) level in the sample or standard.;
Giveaways
Increment services
-
 Single-component Reagents of Assay Kit
Single-component Reagents of Assay Kit
-
 Lysis Buffer Specific for ELISA / CLIA
Lysis Buffer Specific for ELISA / CLIA
-
 Quality Control of Kit
Quality Control of Kit
-
 CLIA Kit Customized Service
CLIA Kit Customized Service
-
 Disease Model Customized Service
Disease Model Customized Service
-
 Serums Customized Service
Serums Customized Service
-
 TGFB1 Activation Reagent
TGFB1 Activation Reagent
-
 Real Time PCR Experimental Service
Real Time PCR Experimental Service
-
 Streptavidin
Streptavidin
-
 Fast blue Protein Stain solution
Fast blue Protein Stain solution
-
 Single-component Reagents of FLIA Kit
Single-component Reagents of FLIA Kit
-
 Streptavidin-Agarose Beads
Streptavidin-Agarose Beads
Citations
- Changes of ubiquitin C-terminal hydrolase-L1 levels in serum and urine of patients with white matter lesionsPubMed: 26232084
- Increased plasma UCH-L1 after aneurysmal subarachnoid hemorrhage is associated with unfavorable neurological outcomePubmed:26810533
- Serum Carnosine Dipeptidase 1 and Ubiquitin C - Terminal Hydrolase L1 as Markers of Brain Damage in Patients after Carotid Endarterectomy mnstemps:135
- Activation of hepatic stellate cells by the ubiquitin C-terminal hydrolase 1 protein secreted from hepatitis C virus-infected hepatocytespubmed:28667290
- Relationships between markers of neurologic and endothelial injury during critical illness and long-term cognitive impairment and disabilityPubmed:29523900
- Serum concentration of ubiquitin c-terminal hydrolase-L1 in detecting severity of traumatic brain injuryarticle:10.1088
- Peripheral blood biomarkers in aneurysmal subarachnoid hemorrhageISBN:978-952-03-0750-9
- Association of neuronal repair biomarkers with delirium among survivors of critical illnessPubmed: 31896448
- BDNF and IL-8, But Not UCHL-1 and IL-11, Are Markers of Brain Injury in Children Caused by Mild Head TraumaPubmed: 32987792