ELISA Kit for Renin (REN) 
angiotensinogenase
- UOM
- FOB US$ 454.00 US$ 648.00 US$ 2,916.00 US$ 5,508.00 US$ 45,360.00
- Quantity
Overview
Properties
- Product No.SEA889Mu
- Organism SpeciesMus musculus (Mouse) Same name, Different species.
- ApplicationsEnzyme-linked immunosorbent assay for Antigen Detection.
Research use only - DownloadInstruction Manual
- CategoryEndocrinologyCardiovascular biologyKidney biomarkerHormone metabolism
Sign into your account
Share a new citation as an author
Upload your experimental result
Review

Contact us
Please fill in the blank.
Recovery
Matrices listed below were spiked with certain level of recombinant Renin (REN) and the recovery rates were calculated by comparing the measured value to the expected amount of Renin (REN) in samples.
| Matrix | Recovery range (%) | Average(%) |
| serum(n=5) | 81-101 | 93 |
| EDTA plasma(n=5) | 78-99 | 83 |
| heparin plasma(n=5) | 91-105 | 95 |
Precision
Intra-assay Precision (Precision within an assay): 3 samples with low, middle and high level Renin (REN) were tested 20 times on one plate, respectively.
Inter-assay Precision (Precision between assays): 3 samples with low, middle and high level Renin (REN) were tested on 3 different plates, 8 replicates in each plate.
CV(%) = SD/meanX100
Intra-Assay: CV<10%
Inter-Assay: CV<12%
Linearity
The linearity of the kit was assayed by testing samples spiked with appropriate concentration of Renin (REN) and their serial dilutions. The results were demonstrated by the percentage of calculated concentration to the expected.
| Sample | 1:2 | 1:4 | 1:8 | 1:16 |
| serum(n=5) | 86-102% | 98-105% | 95-103% | 79-103% |
| EDTA plasma(n=5) | 83-92% | 86-101% | 78-96% | 79-88% |
| heparin plasma(n=5) | 82-105% | 88-99% | 95-104% | 87-95% |
Stability
The stability of kit is determined by the loss rate of activity. The loss rate of this kit is less than 5% within the expiration date under appropriate storage condition.
To minimize extra influence on the performance, operation procedures and lab conditions, especially room temperature, air humidity, incubator temperature should be strictly controlled. It is also strongly suggested that the whole assay is performed by the same operator from the beginning to the end.
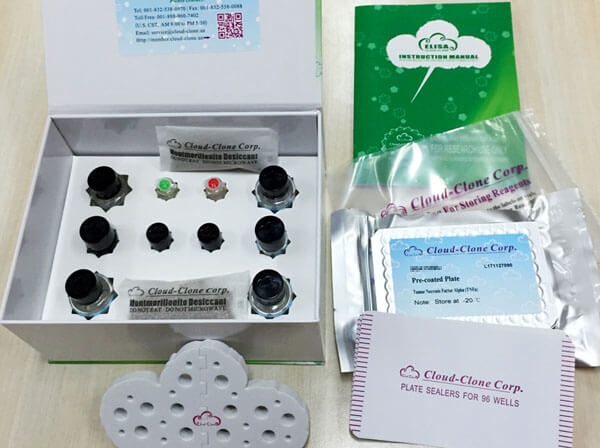
Reagents and materials provided
| Reagents | Quantity | Reagents | Quantity |
| Pre-coated, ready to use 96-well strip plate | 1 | Plate sealer for 96 wells | 4 |
| Standard | 2 | Standard Diluent | 1×20mL |
| Detection Reagent A | 1×120µL | Assay Diluent A | 1×12mL |
| Detection Reagent B | 1×120µL | Assay Diluent B | 1×12mL |
| TMB Substrate | 1×9mL | Stop Solution | 1×6mL |
| Wash Buffer (30 × concentrate) | 1×20mL | Instruction manual | 1 |
Assay procedure summary
1. Prepare all reagents, samples and standards;
2. Add 100µL standard or sample to each well. Incubate 1 hours at 37°C;
3. Aspirate and add 100µL prepared Detection Reagent A. Incubate 1 hour at 37°C;
4. Aspirate and wash 3 times;
5. Add 100µL prepared Detection Reagent B. Incubate 30 minutes at 37°C;
6. Aspirate and wash 5 times;
7. Add 90µL Substrate Solution. Incubate 10-20 minutes at 37°C;
8. Add 50µL Stop Solution. Read at 450nm immediately.

Test principle
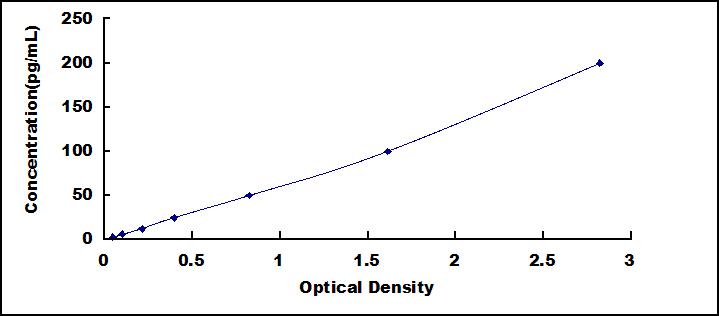
The test principle applied in this kit is Sandwich enzyme immunoassay. The microtiter plate provided in this kit has been pre-coated with an antibody specific to Renin (REN). Standards or samples are then added to the appropriate microtiter plate wells with a biotin-conjugated antibody specific to Renin (REN). Next, Avidin conjugated to Horseradish Peroxidase (HRP) is added to each microplate well and incubated. After TMB substrate solution is added, only those wells that contain Renin (REN), biotin-conjugated antibody and enzyme-conjugated Avidin will exhibit a change in color. The enzyme-substrate reaction is terminated by the addition of sulphuric acid solution and the color change is measured spectrophotometrically at a wavelength of 450nm ± 10nm. The concentration of Renin (REN) in the samples is then determined by comparing the O.D. of the samples to the standard curve.
Giveaways
Increment services
-
 Single-component Reagents of Assay Kit
Single-component Reagents of Assay Kit
-
 Lysis Buffer Specific for ELISA / CLIA
Lysis Buffer Specific for ELISA / CLIA
-
 Quality Control of Kit
Quality Control of Kit
-
 ELISA Kit Customized Service
ELISA Kit Customized Service
-
 Disease Model Customized Service
Disease Model Customized Service
-
 Serums Customized Service
Serums Customized Service
-
 TGFB1 Activation Reagent
TGFB1 Activation Reagent
-
 Real Time PCR Experimental Service
Real Time PCR Experimental Service
-
 Streptavidin
Streptavidin
-
 Fast blue Protein Stain solution
Fast blue Protein Stain solution
-
 Single-component Reagents of FLIA Kit
Single-component Reagents of FLIA Kit
-
 Streptavidin-Agarose Beads
Streptavidin-Agarose Beads
Citations
- Prenatal overexposure to glucocorticoids programs renal 11β-hydroxysteroid dehydrogenase type 2 expression and salt-sensitive hypertension in the ratPubMed: 21045727
- Recombinant human erythropoietin in humans down-regulates proximal renal tubular reabsorption and causes a fall in glomerular filtration rate Physoc: 194241
- Erythropoietin down‐regulates proximal renal tubular reabsorption and causes a fall in glomerular filtration rate in humansPubMed: 20724370
- Anti-Angiotensin and Hypoglycemic Treatments Suppress Liver Metastasis of Colon Cancer CellsKarger: 000330169
- The Angiotensin Receptor Blocker, Azilsartan Medoxomil (TAK-491), Suppresses Vascular Wall Expression of Plasminogen Activator Inhibitor Type-I Protein Potentially Facilitating the Stabilization of Atherosclerotic PlaquesLWW: 08000
- Multiple and large simple renal cysts are associated with prehypertension and hypertension.Pubmed: 23389415
- Estimation of the parameters of the renin-angiotensin-aldosterone system, water-electrolyte metabolism, and endothelial function in newborns of women with chronic hypertensionSpringer:Source
- Organ-Specific Activation of the Gastric Branch of the Efferent Vagus Nerve by Ghrelin in Urethane-Anesthetized RatsPubmed:24366191
- VSL#3 probiotic treatment decreases bacterial translocation in rats with carbon tetrachloride-induced cirrhosisPubmed:24750552
- Immunological and hemostasiological disorders in women with ovarian hyperstimulation syndromePubmed:25200828
- Age-Associated Changes in the Vascular Renin-Angiotensin System in MicePubmed:27200147
- Ablation of Potassium-Chloride Cotransporter Type 3 (Kcc3) in Mouse Causes Multiple Cardiovascular Defects and Isosmotic PolyuriaPubmed:27166674
- Reduced Expression of the Extracellular Calcium-Sensing Receptor (CaSR) Is Associated withActivation of the Renin-Angiotensin System (RAS) to Promote Vascular Remodeling in the Pathogenesis of Essential Hypertension.pubmed:27391973
- Efficacy and Safety of Renal Sympathetic Denervation on Dogs with Pressure Overload-Induced Heart Failure.pubmed:27555054
- Sacubitril/valsartan (LCZ696) Significantly Reduces Aldosterone and Increases cGMP Circulating Levels in a Canine Model of RAAS ActivationDoi: 10.1101/435560
- Gut Dysbiosis Contribute to High Fructose Induced Salt Sensitive Hypertension In Sprague-Dawley Rats
- The vascular endothelial growth factor trap aflibercept induces vascular dysfunction and hypertension via attenuation of eNOS/NO signaling in mice33303990
- Association of Ang-(1¨C7) and des-Arg 9 BK as new biomarkers of obesity and cardiometabolic risk factors in adolescents33568792
- Phosphorylation of β1-integrin in juxtaglomerular cells helps control blood pressure during the progression of diabetic nephropathy
- Bedeutung des intrarenalen Renin-Angiotensin-Systems f¨¹r die Entwicklung der chronischen Nierenkrankheit