Processing of samples for Immunohistochemistry (IHC)
Immunohistochemistry (IHC) is used for identification and localization of the target antigen/antibody by observing the staining results of sections. During the process of sectioning, it is essential to maintain the integrity of antigens. Dissolution and denaturation of antigen should be avoided during the process of staining, washing and embedding. Meanwhile, it should be eliminated that antigens diffuse to near by.
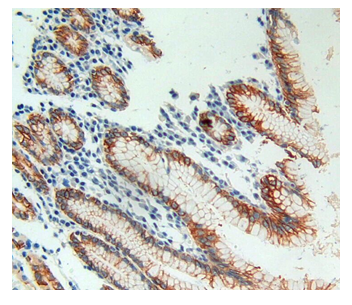
Figure1 IHC image cells or tissue spaces.
Tissue processing is important for obtaining good result of IHC. Living tissue, various body fluid, punctate and cells can be used for IHC analysis. Different process methods can be used according to various specimens. For living tissue, Paraffin tissue sections and frozen tissue sections is usually recommended. Body fluid, punctate and cells are processed by smear method. For bacterial colony or lesion tissues from dead organism, printing slices can be made by pressing fresh section on the slide. Herein we will introduce the common tissue processing technology for living tissue-frozen tissue section and paraffin tissue section.
1.Frozen tissue section
1) Method
①Materials. Select freshly isolated tissue, cut into small pieces of approximately 2mm thickness.
②Quick freezing. Place the tissue piece into a clean box, apply OCT embedding compound to completely cover the tissue. Drop the box into liquid nitrogen and remain there for 10-20s for fast freezing. Store at −80℃ for long-term storage.
⑧Sticking. Put gelatin subbing solution on a clear slide, add a drop of distilled water and paste the sections on the slide. Transfer the slide onto a slide warmer at 35°C, then remove liquid using absorbent paper and dry slide on the warmer.
2) Advantage: a clear preservation of fine of tissue will be obtained.
3) Disadvantage: antigen in tissue will partially lose activity during the preparation of Paraffin tissue section.


